  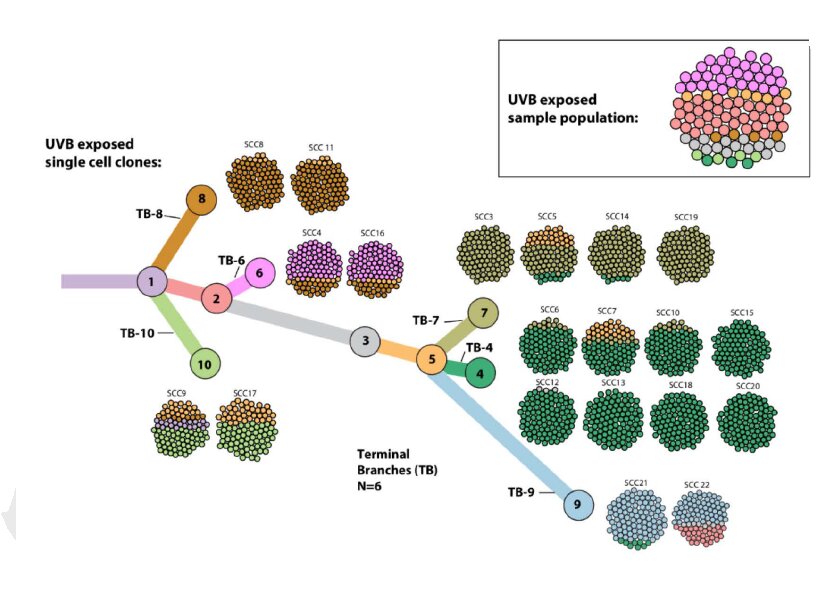
Immune checkpoint inhibitors (ICIs), such as anti-PD-1 (a monoclonal antibody against PD-1), can block co-inhibitory receptors on activated T cells and unleash T cells that have become dysfunctional in the suppressive tumor microenvironment (TME). To prevent uncontrolled immune responses and collateral tissue damage, our immune system develops mechanisms to intrinsically shut down immune responses, which include the expression of co-inhibitory receptors on activated T cells, such as programmed death 1 (PD-1). CD4 T cells are also called helper T lymphocytes, and these cells have many accessory functions (e.g., secreting cytokines or helping B cells to produce antibodies). CD8 T cells can be activated and differentiate into cytotoxic T lymphocytes (CTLs) that can directly kill cancer cells. T cells can be divided into two types: CD8 and CD4 T cells. Our adaptive immune system includes T and B cells. Thus, if successful, immunotherapy can potentially eliminate cancers for a lifetime. In the case of melanoma treatment, some patients are in remission for more than 5 to 10 years. Lastly, immune cells can develop memory responses against cancer cells, which means long-term, durable protection. Also, immune cells have great specificity – they can be trained to recognize and target cancer cells, which means fewer and less serious side effects against other cells in the body. Such an approach is advantageous, because immune cells can attack cancer cells systemically throughout the body, which is particularly relevant in metastatic cancers that can no longer be targeted by surgical intervention. Cancer immunotherapy works by enhancing the powers of the host’s own immune system to fight cancers. Such flexibility displayed by tumors is probably the reason why cancer immunotherapy has become so popular – the adpative immune system can fight tumors’ flexibility with equal adaptability and specificity. Even within the same tumor and the same patient, subclones with different mutations exist, so called intratumoral heterogeneity, which can mean that certain cells can resist treatments, and the cancer can relapse. Intertumoral heterogeneity is frequently observed between patients who often do not respond to the same treatments. Tumor heterogeneity presents a huge challenge when it comes to treating cancer patients. Elucidating mechanistic differences in different individuals’ immune systems will allow us to better harness immune system to design new personalized cancer immunotherapy. Besides tumor-intrinsic heterogeneity and host microbiome, I also discuss the various factors that may constantly shape the dynamic TCR repertoire. This idea may provide a novel perspective to further dissect the mechanisms underlying heterogeneous anti-tumor immune responses in different hosts. This idea implicates that the TCR repertoire in a given patient might not provide sufficiently different TCR clones that can recognize tumor antigens, namely, “a hole in the TCR repertoire” might exist. Here, I propose a provocative concept that intrinsic differences in the T cell receptor (TCR) repertoire of individuals may influence the outcome of anti-tumor immunity by affecting the frequency and/or variety of tumor-reactive CD8 and/or CD4 tumor-infiltrating lymphocytes. The question as to why some hosts can eradicate their tumors whilst others succumb to tumor-progression remains unanswered. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
